Until recently, the most common method for sterilizing spices and bulk food products was through Gamma Irradiation. However, Europe has begun raising significant regulatory difficulties regarding the use of nuclear radiation on food products, and the world is starting to align with this trend. This change reinforces the need for effective and verifiable thermal sterilization solutions, such as SIP.
In process engineering, saturated steam works at high temperatures in a way that optimizes the destruction of proteins and cellular structures of microorganisms. Against this background, steam sterilization is a fundamental and uncompromising requirement in GMP (Good Manufacturing Practice) standards. It is important to distinguish between steam sterilization (moist heat), which achieves faster and more effective results due to the latent heat of condensation, and dry heat sterilization, which requires higher temperatures and longer exposure times.
Scientific Principles and Process Engineering
The success of steam sterilization does not depend solely on temperature, but on a combination of critical factors known as Critical Sterilization Parameters (CIT): temperature, time, and steam quality.
The Principle of Thermal Optimization: The sterilization process requires a critical kinetic balance. A direct relationship exists between the level of thermal exposure (time and temperature) and the logarithmic reduction in microbial load, but this relationship occurs concurrently with the rate of thermal degradation of the product (impacting color, taste, and nutritional values, etc.). Thus, the process is defined as an optimization aimed at achieving the required safety level (SAL) while minimizing harm to the product’s quality attributes.
The Role of Moist Heat: When saturated steam contacts a cold surface (the vessel or the raw material itself within the vessel), it immediately condenses into water, releasing enormous thermal energy (approx. 540 calories per gram) in the process. This thermal energy heats the surface and the raw material itself rapidly and with significantly higher efficiency than heating with dry air. This mechanism is key to the rapid destruction of microbial cellular structures.
Microbial Destruction Kinetics: To ensure an acceptable level of assurance, engineers rely on kinetic concepts:
- D-Value: This is the time required for 90% destruction (a one-log reduction) of a given microbial population at a specific temperature. For example, if the D-Value is 1 minute, it means it will take 10 minutes to reduce the population by ten billion ($10^{10}$).
- Sterility Assurance Level (SAL): In the food industry, especially for sensitive products like spices where dramatic microbial load reduction is needed, the goal is to achieve significant logarithmic reduction of pathogenic organisms and spores. For example, many food preservation processes use a ’12D treatment’ (12-log destruction), ensuring the highest level of safety. Compliance with this required SAL level is the ultimate goal of every sterilization cycle.
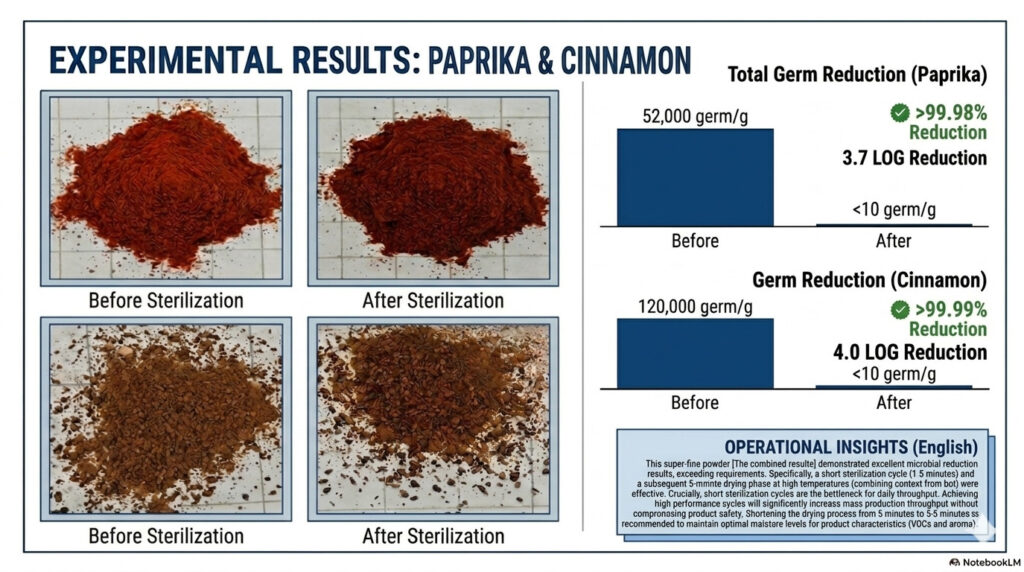
Technology and Equipment (Engineering Applications)
In the food and pharmaceutical industries, two main methods exist for applying steam sterilization, with each having advantages and disadvantages that dictate equipment selection:
- Autoclaves (Autoclaves / Steam Sterilizers): An autoclave is a static pressure vessel primarily used for batch sterilization of components and tools that can be dismantled and transported. Common types include gravity displacement autoclaves and those that use vacuum to remove air and improve steam penetration.
- SIP in the Mixer: The Sterilization In Place (SIP) method is vital for large, fixed production systems, such as dynamic mixing vessels (mixers, reactors). In this method, steam is introduced directly into the equipment, which is pre-designed to withstand the required temperature and pressure for sterilization. The dynamic action of the mixer during SIP is essential for uniform heat distribution and preventing “cold spots.”
- Advantages and Disadvantages of Each Method:

Operational Challenges and Considerations
Achieving successful sterilization in large industrial equipment is an engineering challenge requiring careful planning. In equipment like powder or paste mixers, several critical obstacles exist:
- Condensate Management: The most common problem. Condensed steam turns into water that must be drained. Stagnant water is not only non-sterile but also becomes potential “air pockets” and causes corrosion. Faulty engineering design (e.g., incorrect drainage slopes or lack of efficient steam traps) will compromise SIP reliability.
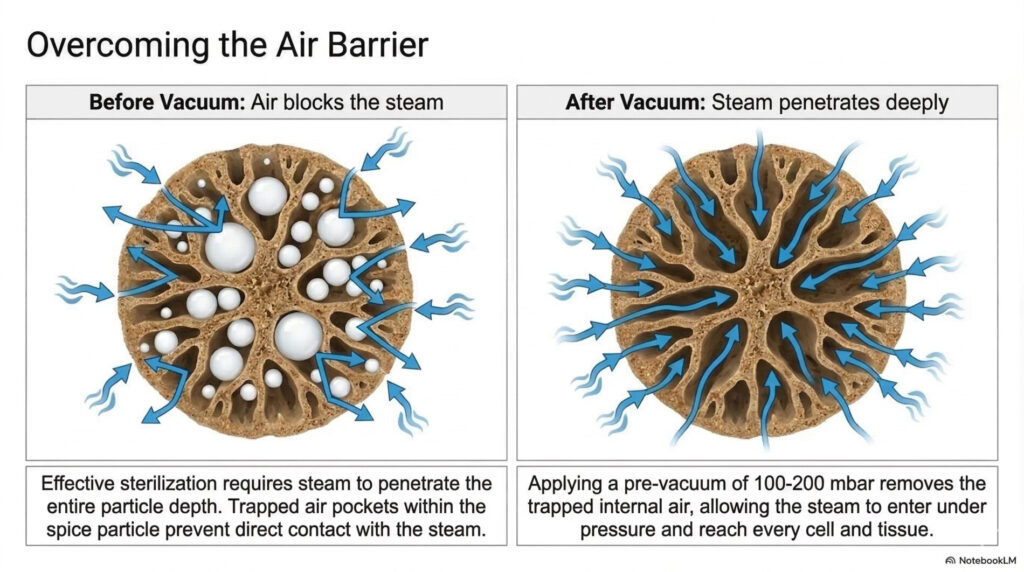
- Air Removal: Air is a non-condensable gas that creates “dry heat pockets” or cold spots that fail to reach the required sterilization temperature (such as 121°C or 134°C). Effective vacuum or purging processes are required to remove all air before steam injection. In complex systems, air is easily trapped in corners, seals, or within equipment components.
- Material Compatibility: Repeated exposure to high-pressure steam at temperatures above 121 C requires durable materials. It must be ensured that all system components—from seals and valves to the vessel’s internal finish—are made of materials that can survive harsh cycle conditions without losing integrity or releasing unwanted particles into the product. Selecting appropriate seals (such as EPDM or PTFE) is particularly critical.
- Energy and Cycle Time Considerations: Sterilization is a costly process in terms of energy (heating and steam generation) and time (waiting for cooling). Correct design of the equipment and process can shorten the cycle time, which directly improves production efficiency.
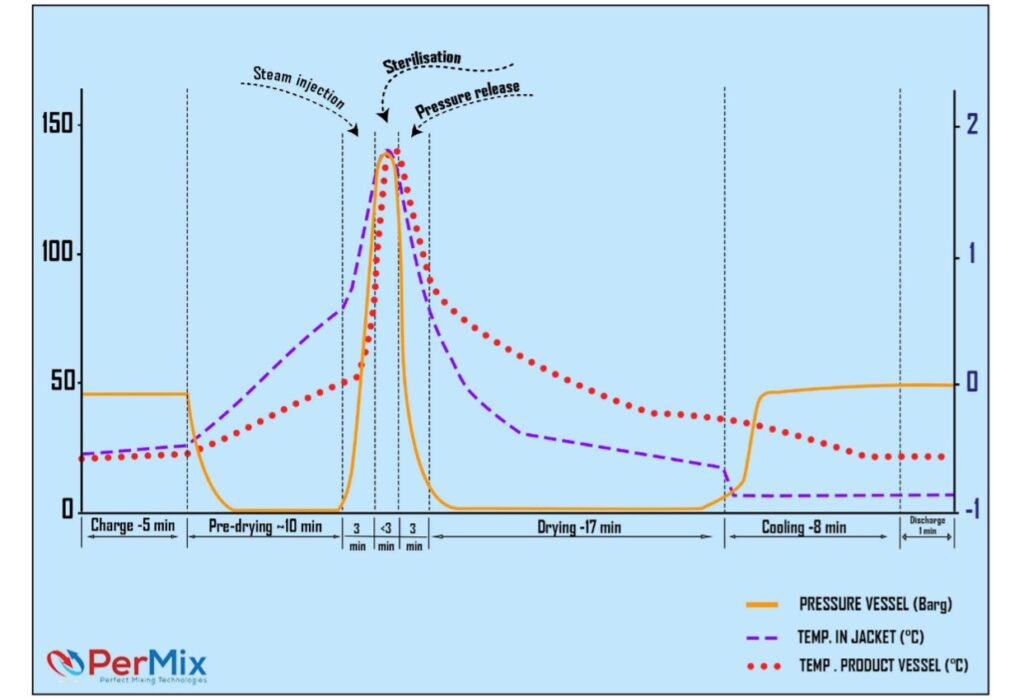
Summary and Future Trends
Steam sterilization remains a key technology in modern industry. Success in this process requires a winning combination of clear scientific principles (D-Value and SAL) and precise engineering design (SIP, condensate drainage, and air management).
As regulatory demands become more stringent, the importance of expert equipment manufacturers designing mixing systems and SIP/CIP (Cleaning In Place) procedures that are verifiable and reliable from the outset grows. The design of the mixing system must account for sterilization requirements from the start, while avoiding dead legs or poor drainage points.
Future Trends: Despite the clear advantage of steam, technological developments offer alternatives, such as sterilization using vaporized hydrogen peroxide (VHP). Nevertheless, steam continues to maintain its position as the undisputed technology for large process vessels, as it is cheaper, safer, and based on a natural process. The future focus is on improving cycle efficiency and the reliability of online monitoring of critical parameters.














